Geistlich Derma-Gide®

Advanced Design
Advanced Wound Matrix
Geistlich Derma-Gide® Second generation xenograft featuring 4 dimensions of product design:
- Dual Sourced
- Highly Refined
- Bi-Layered
- Structurally Optimized
Discover how the advanced design of Geistlich Derma-Gide® transforms two distinct source tissues to a clinically effective Advanced Wound Matrix.


- Two distinct sources from porcine are selected for their specific characteristics and provide the best composition for further refinement.
- These two source tissues are protected by trade secrets and are not disclosed to the public but are not SIS or dermis.

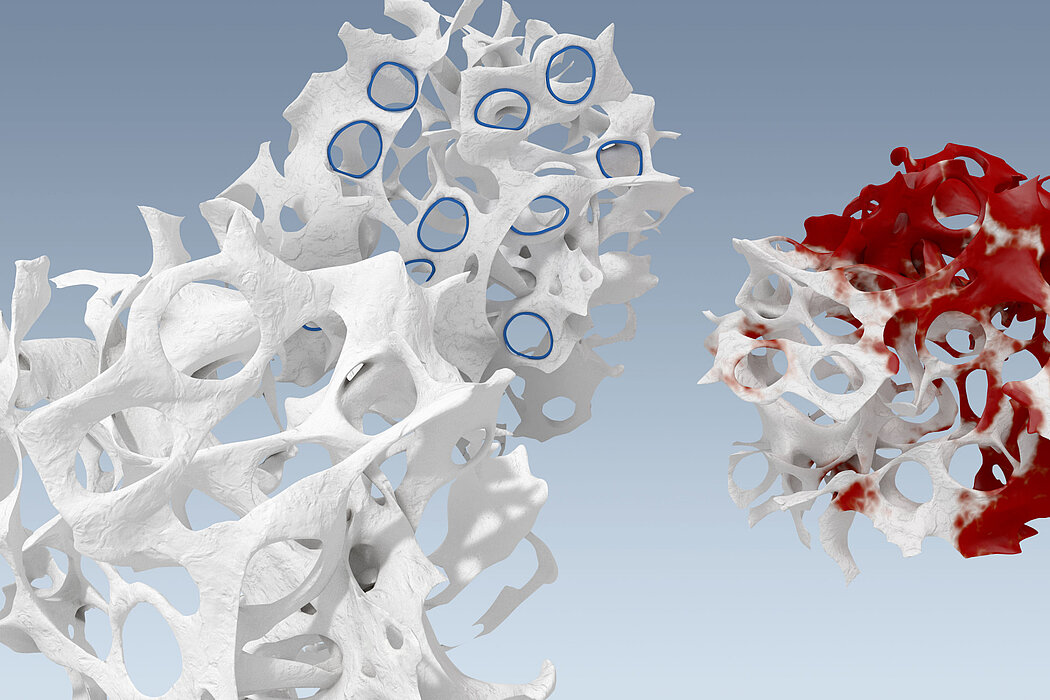
- Based on 170 years of expertise, Geistlich transforms the source tissues into an advanced design with the precise characteristics to support wound healing.
- The refinement step is what differentiates Geistlich Derma-Gide® from allografts - which can only be minimally manipulated – and other first generation xenografts.

Geistlich Derma-Gide® features a unique bilayer design.
01/05
- The bi-layered design of Geistlich Derma-Gide® mimics the structural organization of human skin facilitating re-epithelialization and cell ingrowth.
- The integrated structure is a result of the refinement process and the two layers are mechanically linked to form a durable and easy to use solution.


- The upper compact layer of Geistlich Derma-Gide® mimics the skin’s basement membrane allowing keratinocyte migration as well as growth factor binding and preservation. It also protects the wound site and allows suturing if desired.
- The structure of the lower porous layer of Geistlich Derma-Gide® supports MMP activity modulation which supports the formation of a physiological extracellular matrix. It also provides an optimized environment for cell migration, attachment and vascularization and can absorb up to nine times its weight in wound fluids.
CAUTION: This product is only available in the U.S.A. Federal (U.S.A.) law restricts these devices to sale by or on the order of a physician.




